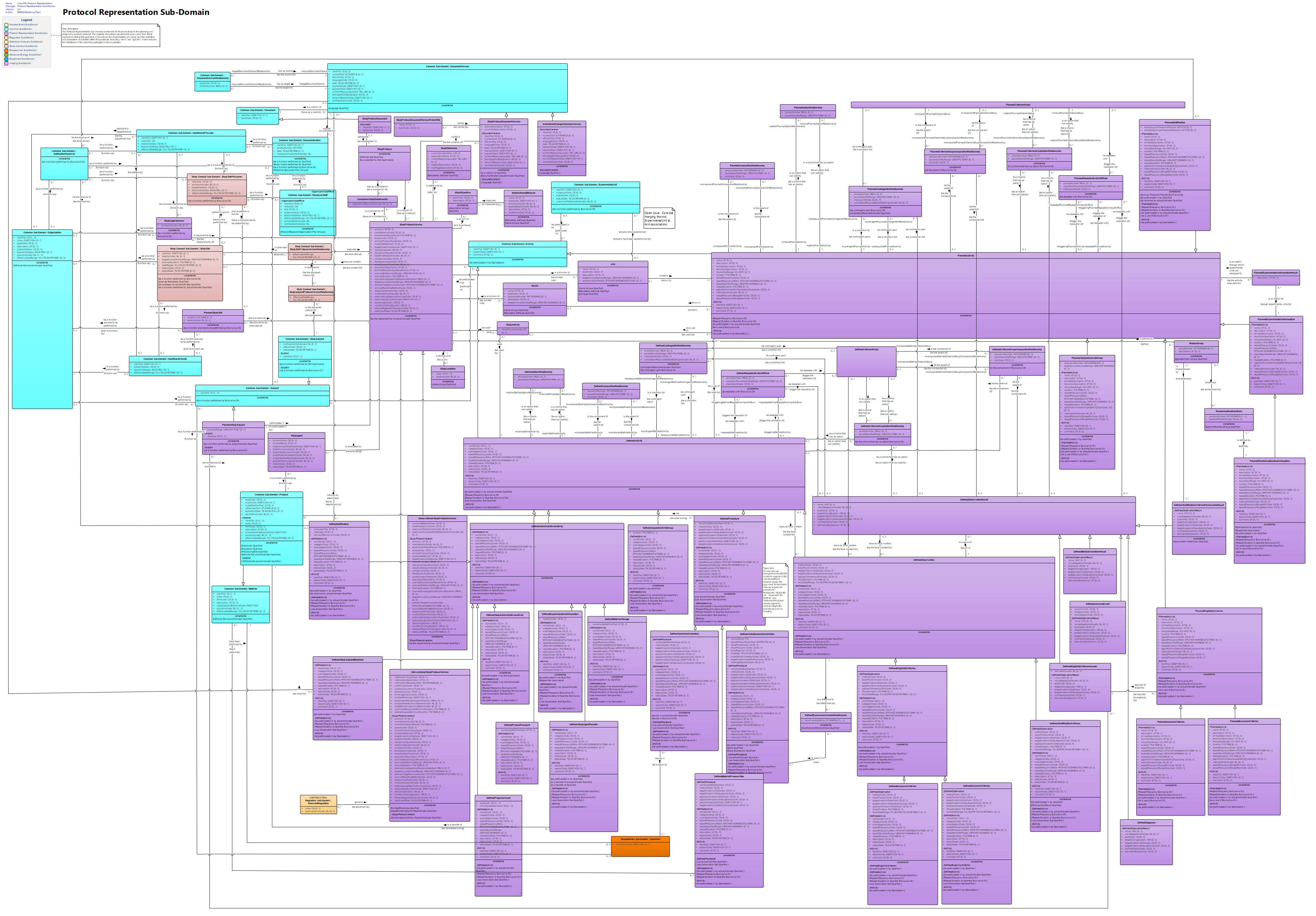
View PR: Protocol Representation : Class diagram
| Created: | 6/1/2009 12:00:00 AM |
| Modified: | 8/5/2019 12:27:45 PM |
 Project: Project: |
|
 Advanced: Advanced: |
The Protocol Representation sub-domain is intended for those involved in the planning and design of a research protocol. The majority of business requirements have come from those involved in clinical trial protocols. It focuses on the characteristics of a study and the definition and association of activities within the protocols, including "arms" and "epochs". It also includes the definitions of the roles that participate in those activities.<br/>